The Chemistry of Pigments: Why Paints and Dyes Behave Differently
Pick up a tube of oil paint and a bottle of fabric dye. One feels thick and opaque; the other is thin and vibrant. Apply them to a surface, and the differences become even clearer: the paint covers solidly and lasts for decades, while the dye soaks deep into fibers, creating rich but sometimes more delicate colors.
The reason isn’t just how we use them — it’s rooted in their fundamental chemistry.
Pigments: Insoluble Particles That Sit on Top
Pigments are solid, insoluble color particles. They do not dissolve in the medium (water, oil, acrylic binder, etc.). Instead, they are suspended or dispersed throughout it.
- Particle size: typically 0.1–10 micrometers— large enough to scatter light.
- How they work: The binder (oil, acrylic polymer, gum arabic) holds the pigment particles in place on the surface of the material.
- Key properties:
- Opacity: Excellent light-scattering ability (especially inorganic pigments like titanium dioxide).
- Durability: Generally more lightfast and chemically stable.
- Hiding power: One or two coats can completely cover the underlying surface.
Common examples:
- Titanium dioxide (TiO2)—the most widely used white pigment in paints due to its high refractive index.
- Iron oxides—earth tones in artist paints.
- Phthalocyanine blues and greens — modern organic pigments.
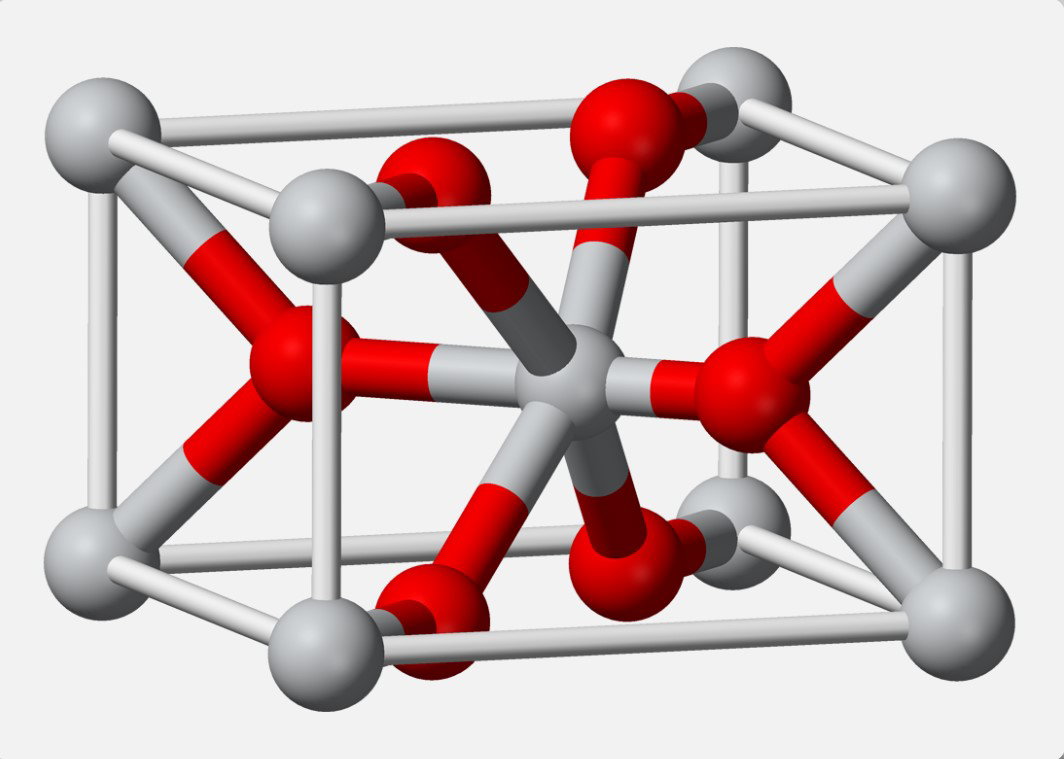
Caption: Crystal structure of titanium dioxide (TiO₂), a classic inorganic pigment prized for its opacity and brightness in paints.
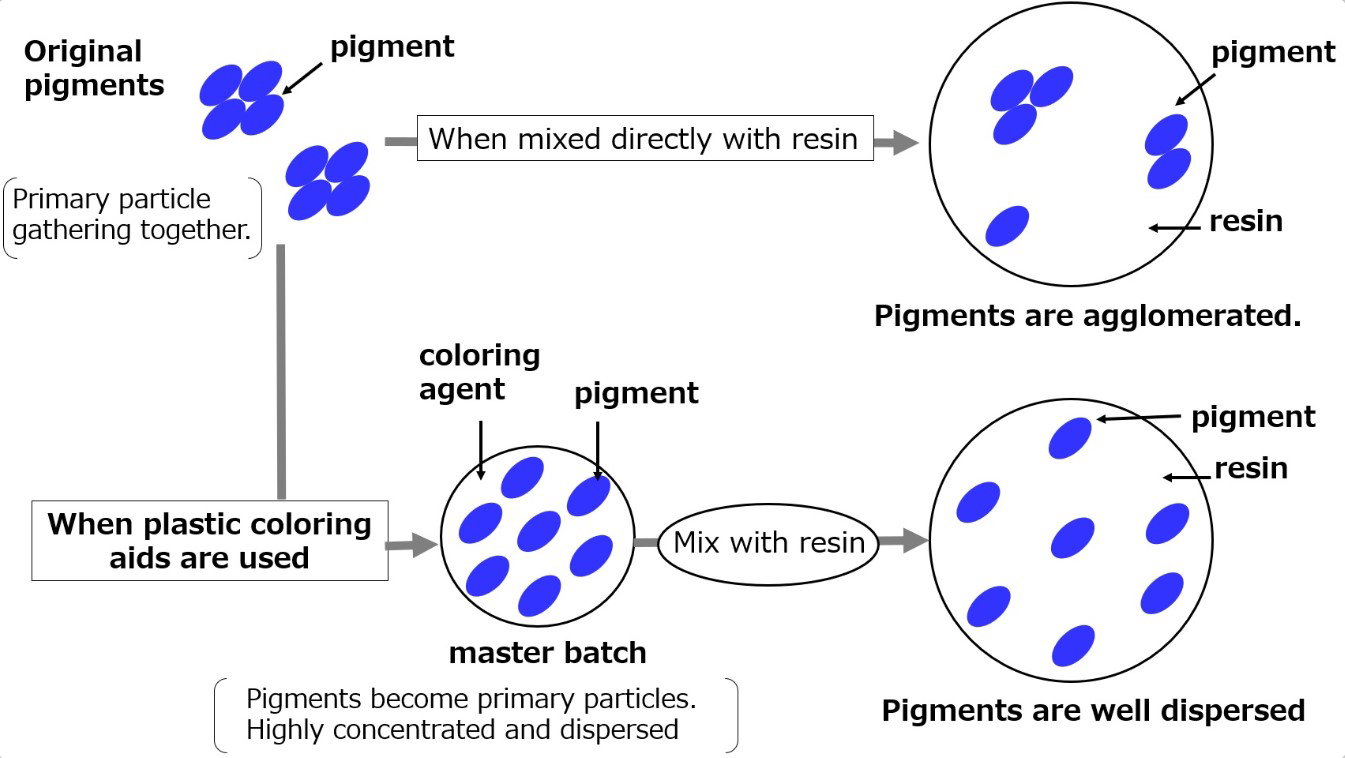
Caption: Pigment particles are insoluble and must be properly dispersed in a binder. Without dispersants, they can clump (agglomerate), reducing color strength.
Dyes: Soluble Molecules That Penetrate and Bond
Dyes are organic molecules that fully dissolve in a solvent (usually water or alcohol). They are tiny—on the molecular level— and interact chemically with the substrate.
- How they work: Dye molecules penetrate the material (fabric fibers, leather, paper) and form chemical bonds (ionic, hydrogen, or covalent) with it.
- Key properties:
- Transparency / Translucency: They don’t scatter light much; instead, they absorb specific wavelengths and let others pass through.
- Vibrancy: Often produce more intense, luminous colors.
- Fastness: Can be less lightfast or washfast unless specially fixed (mordants or reactive groups help).
Common examples:
- Azo dyes — bright reds, yellows, and oranges used in textiles.
- Anthraquinone dyes — vibrant blues and greens.
- Natural dyes like indigo or cochineal.

Caption: Clear comparison — Dyes dissolve and chemically bond with materials, while pigments remain as insoluble particles held by a binder.
Why They Behave So Differently: Key Chemical Differences
| Property | Pigments | Dyes |
|---|---|---|
| Solubility | Insoluble in the medium | Soluble in water or solvent |
| Particle / Molecule Size | Larger particles (microns) | Molecular scale |
| Light Interaction | Scatter + absorb light (opaque) | Primarily absorb light (transparent) |
| Application | Sit on surface with the binder. | Penetrate and bond inside substrate |
| Lightfastness | Usually higher (especially inorganics) | Often lower unless fixed or reactive |
| Best For | Paints, inks, plastics, coatings | Textiles, leather, food coloring, inkjet inks |

Caption: Lightfastness testing shows how some pigments and dyes fade differently when exposed to light over time.
Real-World Implications
- Artists & Painters: Choose pigments for opacity and longevity (oil/acrylic paints). Lake pigments (dyes precipitated onto a base) combine some dye vibrancy with pigment stability.
- Textile Industry: Dyes are essential for deep penetration and washability in fabrics. Reactive dyes form strong covalent bonds with cotton.
- Printing & Inks: Pigment-based inks offer better lightfastness for fine art prints; dye-based inks produce brighter colors on photo paper but may fade faster.
- Modern Innovations: Hybrid “pigment dyes” and nanoparticle pigments aim to merge the advantages of both types of dyes.
Final Takeaway
The next time you admire a bold, opaque red in an oil painting versus a vivid, flowing red on silk, remember: you’re seeing two entirely different chemical strategies at work.
Pigments protect and cover by staying put as tiny solid particles.
Dyes seduce and penetrate by dissolving and bonding on a molecular level.
Understanding this chemistry helps artists choose the right materials, designers predict how colors will behave, and manufacturers create longer-lasting, more vibrant products.
Color isn’t just about hue—it's about how the chemistry behind it interacts with light, surface, and time.